
Transformational Science
Qu’s Site Specific Immunomodulators (SSIs) aren’t designed to wage war against disease. They are designed to restore health and normal immune function and transform the way we think about the treatment of cancer, chronic inflammatory diseases, and metabolic disease.
Dr. Hal Gunn, CEO
Addressing the Underlying Causes of Disease
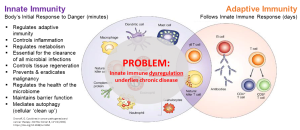
The innate immune system plays a fundamental role in maintaining health and clearing disease. Its important functions include controlling inflammation, modulating adaptive immunity, maintaining barrier function, regulating metabolism, regulating the microbiome, clearing dead and dying cells, and clearing cancer and infection.
Innate immune dysfunction underlies cancer and chronic inflammatory diseases, and this sets the stage for worsening chronic unproductive inflammation and a vicious cycle of further impairment of innate immunity that contributes to disease progression.
Qu’s transformative platform, Site Specific Immunomodulators (SSIs), is uniquely designed to restore normal function of the cells, pathways, and roles of innate immunity in a targeted diseased organ to clear and prevent cancer, chronic inflammatory disease, metabolic disease, and chronic infection. In doing so, Qu’s SSIs are a fundamentally different way to treat and prevent disease that is aligned with the body’s healing processes. Rather than forcing a single pathway of immune function in one direction (i.e., its complete suppression or forced activation) like most immunotherapies, SSIs are designed to restore all the roles and functions of innate immunity, immune balance, and health.
Innate Immune Training
The innate immune system must be ‘exercised’ by occasional exposure to infectious challenges to function optimally, similar to the way that regular physical exercise ensures that muscles maintain strength and function. Without innate immune training, our innate immune system becomes less resilient, loses immunological “tone”, and becomes more susceptible to dysregulation, which can lead to the development of cancer, chronic inflammatory diseases, metabolic disease, and chronic infection.
Qu Biologics’ SSIs are uniquely designed to activate the production of newly trained innate immune cells (through myelopoiesis) and direct (through organ-specific chemokine release) these activated innate immune cells (NK cells, M1 macrophages, etc.) to a targeted disease organ, replacing dysregulated innate immune cells within the diseased organ, and restoring the ability of the immune system to clear disease.
Tailored and Targeted
The immune system evolved to effectively and efficiently clear acute infection. Qu’s pivotal discovery is that the innate immune response to acute infection includes an organ-specific recruitment of activated innate immune cells to the organ in which that bacterial species is endogenous (i.e., lives in the organ’s microbiome) and/or has caused infection previously. Furthermore, Qu discovered that this mechanism can be harnessed without the risk of infection using SSIs (each of which is derived from an inactivated single bacterial species that is a common cause of infection in the targeted organ). SSI treatment efficiently, effectively, and safely activates this natural mechanism that evolved to direct an organ specific recruitment of activated ‘trained’ innate immune cells to the targeted organ. This immunological mechanism evolved to create an efficient, safe, and targeted response to acute infection and then co-evolved to support the prevention and clearance of cancer, chronic inflammatory diseases, and metabolic disease by restoring innate immune function.
The simplest way of thinking about Qu’s SSIs are as ‘innate immune vaccines’ that are designed to restore all the cells, pathways, and functions of innate immunity safely and effectively in a targeted diseased organ.